WEEKLY HIGHLIGHTS 2026 FIRST HALF
HIGHLIGHTS FOR WEEK OF 23 FEBRUARY – 1 MARCH 2026
Impermanence
I have recently noted a paradox. The happier I feel, the more I am afraid of dying.
The opposite is also true. This perhaps explains why people living in desperate circumstances may be more ready to lose their lives (?), or why people who suffer from debilitating diseases are less afraid of dying.
As such, when seeking happiness, there is also a need to become comfortable with dying even during times when we feel happy.
I recently listened to a talk by Jack Kornfield, a buddhist monk and writer. In his talk, Jack Kornfield discusses impermanence and reminded me that we live in a world where nothing is permanent. The talk prompted me to try to embrace the idea of impermanence in my daily living.
He told the story of a Buddhist monk, who held a beautiful Chinese vase in his hand and exclaimed “For me, this vase is already broken.” With his words he expressed the truth that one day the vase will surely break, and that it would be futile to try to hang on to the thought of owning the vase for good. Nonetheless, despite knowing that the vase will break one day, he can still enjoy and cherish the vase in the present moment.
How often in our lives do we try to secure our material possessions, or are determined not to lose our physical fitness. I think there is nothing wrong with valuing material possessions and physical fitness in the moment, but we should do so without being afraid of losing them. Or else, as one student told me in our one-to-one zoom conversation, our material possessions have us. Enjoying to have things in the moment does not mean that things have to stay with us forever.
The desire to maintain things we have is probably rooted in our need to strive for certainty. It seems that most people even prefer to be certain about negative things that might happen to us over being uncertain about the future.
However, certainty is often an illusion. More likely than not, things will be turn out entirely different from what we expect them to. Even in cases where the facts may be certain, it is impossible for us to predict how we will feel about these facts and deal with them in the future. As such, it seems to be a waste of time and energy to try to spend too much effort in order to ensure a certain future.
A much better strategy may to be to focus on the moment and on what we care about now. Our efforts will likely lead somewhere. This approach seems much more authentic and genuine than directing all our efforts towards an imaginary future.
All this also applies to the ultimate uncertainty of when I am going to die, which in fact could be today, tomorrow, or in many years to come.
In an interesting New York Times interview, author George Saunders talked about our perception of our own uniqueness. In other words, we view ourselves as the central player in a movie that assigns all other people to supporting roles.
He suggests that freeing ourselves from the idea that without us, things will not be the same, is liberating.
It is indeed true that when I listen to news of people who have died or who are dying, even though I try to imagine myself in their shoes, I fundamentally believe that this cannot happen to me and that I am different.
However, it is certain is that one day this illusion will be taken from me. It would be good to accept this fact now.
I recently listened to an interesting BBC podcast about a Dutch couple who chose to die together by assisted suicide, which in the Netherlands is referred to as duo-euthanasia. The husband suffered from chronic pain, the wife from early dementia.
What struck me was how the husband said that he is ready to die because he has lived his life.
The reason this struck me is because I feel that we must strive to achieve a state where we can say that we have lived our life and have achieved what is most meaningful to us. Not having achieved this is for me what makes me fear death.
If I look back at the things that I have achieved (of which there aren’t that many), the one that truly satisfies me the most is the personal improvement I have achieved.
I feel that the more I have achieved personal improvement to become the person I want to be, the more I am ready to die.
My biggest achievement of all is to be at peace with myself as a result of having found what makes me most happy and avoiding those things that don’t. It is as though this was the race that I was running all my life, to try to seek what gives me happiness and to overcome the obstacles that prevent me from living a happy and productive life. And the purpose of my life was not the result of achieving that, but the journey there.
HIGHLIGHTS FOR WEEK OF 16 – 22 FEBRUARY 2026
How to maintain happiness when the routine sets in
The semester is in full steam and the daily routine has set in again. There are lots of things to do, some of which aren’t things that I always enjoy or look forward to.
Adding to this, getting older comes with its own challenges like some chronic pain and joint problems. I also find myself having to take a lot more breaks than I used to.
Finally, I also feel a lot of uncertainty about whether things I have planned will work out and worry sometimes that unexpected things might happen.
How to maintain a sense of happiness on a daily basis under these conditions?
Firstly, it is crucial to take action, because feeling happy doesn’t just happen on its own, at least in my experience. On the other hand, in many years of seeking to live a happy life I have also realised that whatever I try, there is no guarantee that I will achieve happiness, even if I am doing things that have previously made me feel happy. As such, all I can do is to create conditions to experience feelings of happiness.
This in turn requires that we find out what has the greatest potential to make us happy and what impedes our ability to experience happiness. We then need to prioritise to make time for the things that potentially make us feel happy, and find ways to eliminate the things that don’t. Both of these are equally important.
All this is easier said than done. On the other hand, gratification does not only come from achieving the end goal (finding happiness). The process of finding ways to achieve the goal, by exploring what things make us feel happy and discovering new ways to eliminate time spent on things that do not bring us happiness or prevent us from feeling it, can be very exciting and rewarding as well.
However, in this post I want to talk about something else I can do – accepting things and changing the way I look at tasks that I have committed to.
Firstly, it is worth reminding myself of how fortunate I am! For the most part of my days, I have the freedom to plan my time in the way I want to and the opportunity to do meaningful things. In my personal life, I am able to do the things I want to without having to think about major financial constraints and above all, I have the privilege to live and work in a beautiful country.
I must say that I regularly do acknowledge how privileged I am (in fact almost every day). But naturally, that by itself is not sufficient to feel happy.
Moreover, it may not be helpful to attach ourselves too much to the privileges we have because there is the possibility that they are taken away from us at any time. And there are people who manage to feel a sense of happiness and meaning despite living in far more difficult circumstances than I do. The opposite, of course, is also true.
This is why the mindset is important. There are always two ways to look at tasks: as something we have to do, or as something we want to do.
However, doing the latter often seems difficult. What makes it difficult? What is it that makes a task uncomfortable or dreading?
This is an important question to ask ourselves. For me, what makes a task uncomfortable or dreading is almost always a sense of uncertainty.
A good example are my running sessions. If I have planned an exciting or fun workout, I look forward to a session. I also look forward if I feel certain that the workout is not too hard and not too easy. For instance, there are times when I try the workout myself before our training session, allowing me to adjust the workout intensity and feel certain about the degree of difficulty. This greatly helps me reduce my anxiety and increase my excitement about training sessions. However, due to injuries or my own training priorities, it has not been possible to do this routinely.
Another example is the student symposium (Young Scientists’ Symposium) I organise every year around this time. If there is something I have prepared (such as a speech) that I feel excited about, I tend to look forward to the event. If not, I tend to perceive the symposium as an emotional burden that affects my sense of happiness.
The same is true in many areas of my professional life, such as my classes or my student one-to-one conversations. If I have devised exciting activities for a class or feel emotionally well-prepared for my student conversations, I look forward to them.
As such, it is best to face the uncertainty and anxiety I experience about upcoming tasks, by taking action to prepare and come up with exciting ideas. And if it is not immediately clear how to come up with these ideas, we have to search for ways! There is a solution to any problem, if only we take the time to find it.
HIGHLIGHTS FOR WEEK OF 9 -15 FEBRUARY 2026
Wings Cross Country Championships 2026
This Saturday, our staff running group saw two teams, our Men’s Open and Men’s Masters teams, compete in the Wings Cross Country Championships 2026 at Bedok Reservoir. For our runners, it was a great experience to compete with the running elite in Singapore. Unexpectedly, despite competing against the best clubs in Singapore, our Masters team even won the Bronze medal, which made it an even more memorable event!
HIGHLIGHTS FOR WEEK OF 2 – 8 FEBRUARY 2026
Activating the NRF2 transcription factor
Many years ago I was greatly inspired by a research paper by Gerald Shulman’s group at Yale. His lab developed a liver-selective mitochondrial uncoupler drug. His group developed a prodrug that is normally inactive, but can be converted to an active uncoupler by drug-metabolising cytochrome P450 enzymes, which are exclusively expressed in the liver. As a consequence, the active uncoupler only accumulates in liver tissue, thus avoiding on-target and off-target side effects in other tissues.
Most new ideas are inspired by other ideas. Hence, being fascinated by the uncoupler prodrug idea, I tried to come up with related research questions. For instance, one project that Ying Yee, an amazing UROPS student in our lab, conducted several ago was to develop a novel prodrug delivery system to kill tumor cells.
A major problem in conventional cancer therapy is dose-limiting drug toxicity. Although most anti-cancer drugs have some selectivity towards cancer cells, this selectivity is usually only partial and they commonly also affect normal cells to some degree. As a result, the doses of anti-cancer drugs that can be given to patients are often limited.
In Ying Yee’s project, we utilised 2-methyl-antimycin A as the prodrug of the cytotoxic complex III inhibitor antimycin A. The end goal of her project was to engineer a cytochrome P450 (CYP450) enzyme that can metabolically activate 2-methyl-antimycin A. The engineered enzyme could potentially be targeted to tumours via cell-based or adeno-associated virus (AAV) delivery systems.
Before trying to engineer such an enzyme, we had to ensure that 2-methyl-antimycin A is not cytotoxic before its conversion, as well as that endogenous human CYP450 enzymes in the liver do not activate 2-methyl-antimycin Ying Yee was able to confirm both of these prerequisites. She also found that expression of CYP450 enzymes in non-hepatic cells is in principle sufficient to mediate prodrug activation, without the need of cotransfecting CYP450 reductases, which appear to be expressed at sufficient levels in non-hepatic cells.
Sadly, engineering a CYP450 enzyme that could activate 2-methyl-antimycin A proved more difficult than expected. And when Ying Yee’s UROPS project ended, the project stopped there, too.
However, another attempt to establish a cytochrome P450-mediated prodrug system proved more successful, and this is the subject (in part) of our most recent publication.
In the project, Mei Ying, who used to be a PhD student in our lab and now is a postdoc at Dana-Farber Cancer Institute, tried to find new prodrugs that could be activated in a liver-selective manner.
In our study, we focussed on the transcription factor NRF2 (short for Nuclear factor-erythroid 2-Related Factor 2). NRF2 has been shown to have beneficial effects in various human diseases and conditions. These include fatty liver disease and its more advanced form, nonalcoholic hepatosteatitis (NASH), which in addition to fat accumulation is characterised by inflammation of liver tissue.
Fatty liver disease and NASH are not only extremely common, they are also worrying because they often progress to liver cirrhosis, liver failure and liver cancer. Furthermore, they are often associated with insulin resistance.
NRF2 functions to induce the expression (via transcriptional activation) of different target gene families, including drug-metabolising enzymes, anti-inflammatory factors as well as enzymes involved in scavenging or detoxifying reactive oxygen species. Both anti-inflammatory and antioxidant mechanisms are likely to contribute to the beneficial effects of NRF2 activation in NASH. As such, many research groups and pharmaceutical companies have been trying to find new activators of NRF2.
Indeed, numerous NRF2 activators have been identified and developed, most of which work by inhibiting its negative regulator, the E3 ubiquitin ligase KEAP1.
Yet, the therapeutic strategy of NRF2 activation has proved difficult to translate into the clinic. This is because activating NRF2 can also have unwanted negative consequences. As we wrote in our original paper:
“… NRF2 activation is known to promote tumorigenesis as well as mediate chemoresistance. Inactivating mutations in KEAP1 or activating mutations in NRF2 are frequently found in a number of cancers, including cancers of the lung, gallbladder, and liver. While generally cytoprotective, NRF2 activators can also exert important other adverse effects. For instance, NRF2 activation has been reported to promote hypertension in diabetic mice via the transcriptional induction of angiotensin and angiotensin-converting enzyme in renal proximal tubule cells. Nrf2 activation in regulatory T cells has been shown to promote regulatory T cell loss and to induce an autoinflammatory phenotype in mice. NRF2 has also been reported to play an important role in regulating hematopoietic stem cell quiescence. In addition, electrophilic NRF2 activators are also likely to have off-target effects by reacting with other target proteins with reactive cysteine residues.”
What these studies suggest is that long-term treatment with NRF2 inducers can promote tumor formation, as well as lead to unwanted side effects in different tissues. Indeed, a clinical trial for the treatment of patients with type 2 diabetes and chronic kidney disease with the Nrf2 activator drug bardoxolone methyl was interrupted prematurely because of an increased incidence of adverse cardiovascular events and death in trial participants.
As such, the challenges associated with developing NRF2 activators are similar to mitochondrial uncouplers, which also have great potential in the treatment of obesity and fatty liver disease, but which at the same time can cause toxic side effects.
Hence, in our project, Mei Ying set out to identify compounds that are inactive in inducing NRF2 activation on their own but that can be activated by hepatic CYP450 enzymes. And it was a very exciting moment when she succeeded to find a number of such compounds!
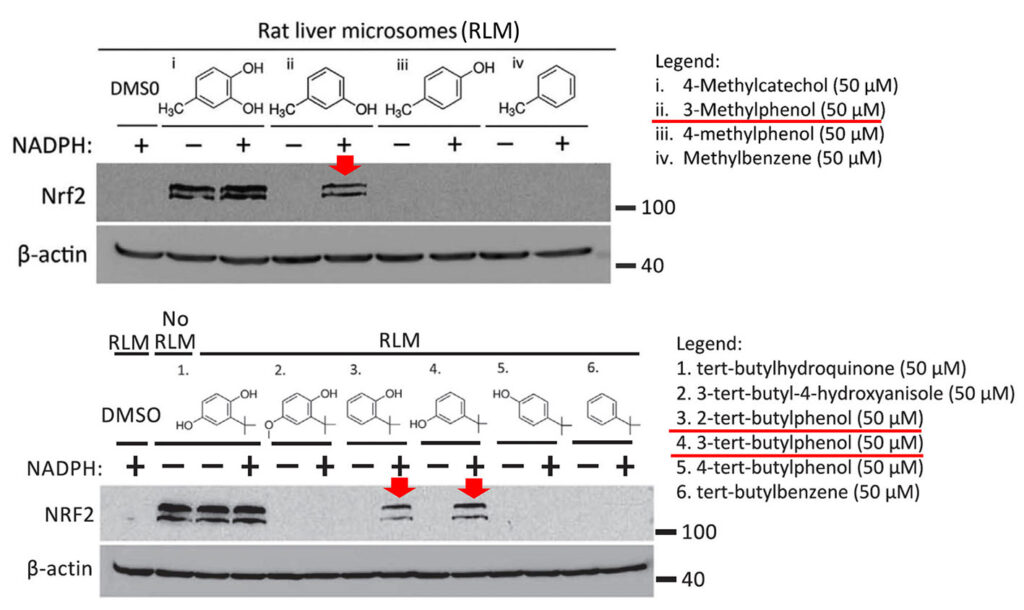
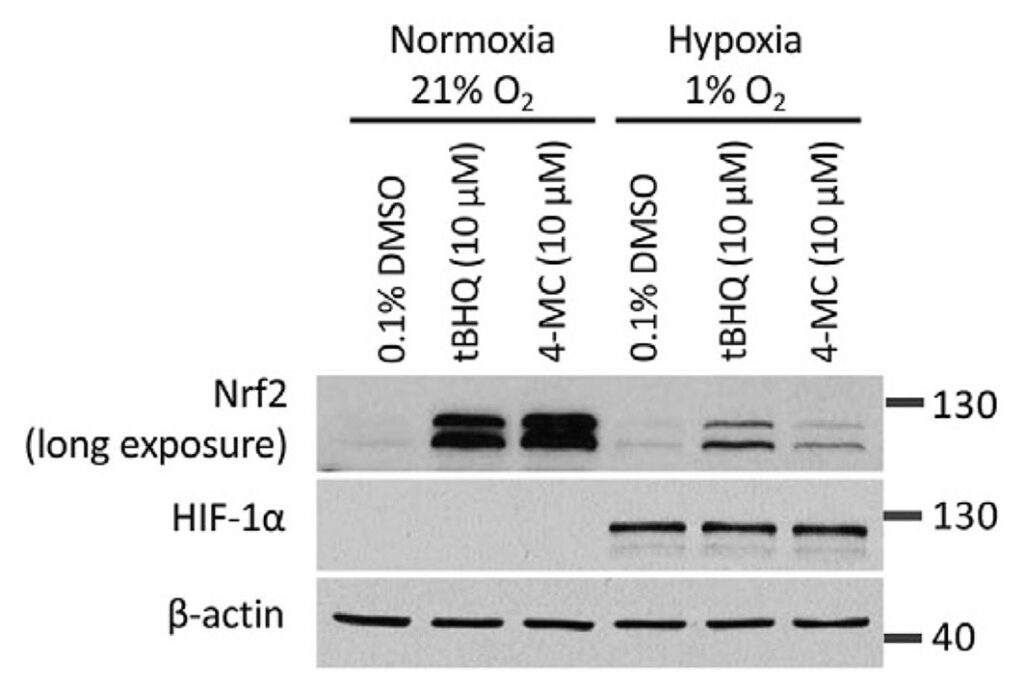
Specifically, as shown in the figure below, Mei Ying found that three compounds, 3-methylphenol, 2-tert-butylphenol and 3-tert-butylphenol, induced the expression of NRF2 after preincubation with rat liver microsomes (i.e. liver endoplasmic reticulum particles that contain CYP450 enzymes). These compounds were inactive without prior activation (not shown in the figure). Moreover, the prodrugs were only activated when the CYP450 co-substrate NADPH was added during the preincubation with liver microsomes.
The three prodrugs are converted into the active compounds 4-methylcatechol (4-MC) and tert-butylhydroquinone (tBHQ), respectively. As expected, the figure also shows that 4-MC and tBHQ themselves activate NRF2 without requiring NADPH-dependent preactivation.
In the shown experiments, HEK293T cells were treated for 4 h with the known NRF2 activator 4-methylcatechol (top panel (i)), or the indicated prodrug analogs of 4-methylcatechol (ii, iii, and iv). In the bottom panel, cells were treated with another NRF2 activator, tert-butylhydroquinone (1) or its prodrug candidates. Prior to the cell treatment, all drugs were preincubated with rat liver microsomes (containing CYP450 enzymes) in the presence or absence of the CYP450 co-substrate NADPH. The results suggest that the prodrug candidates indicated with the red arrows were converted to their active metabolites.
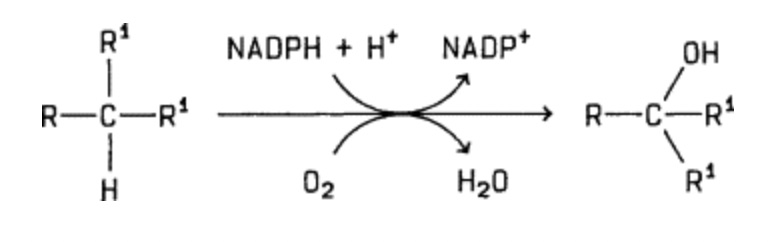
Cytochrome P450 enzymes utilise oxygen as a co-substrate. Given that our experiments were conducted at an atmospheric oxygen concentration of 21%, one valid question is whether the CYP450-mediated prodrug activation also occurs under physiological conditions in vivo, where the oxygen concentrations are much lower. For instance, the physiological oxygen concentration in the liver is only around 4%.
General reaction of CYP450-catalyzed hydroxylases to catalyse substrate hydroxylation
While we did not address this question in our study, it appears likely that the reaction does happen in vivo, given that CYP450 enzymes normally operate under these conditions. Indeed, CYP450 enzymes are known to have a rather high affinity for oxygen.
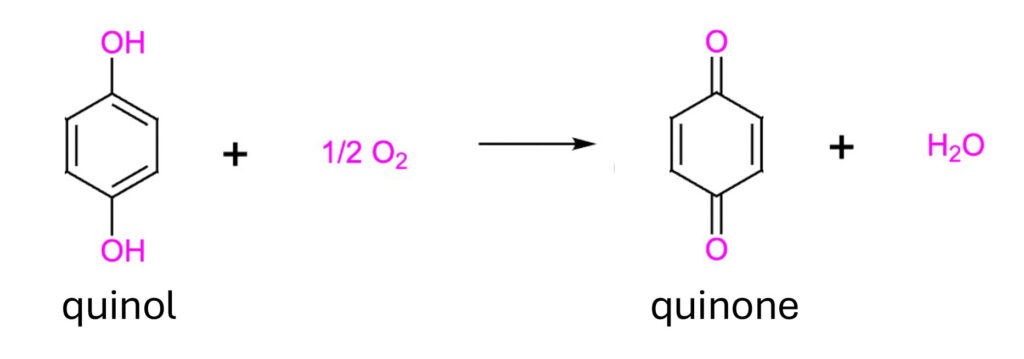
However, more significantly, upon activation of our prodrugs via hydroxylation, the activated quinol compounds (4-methylcatechol (4-MC) and tert-butylhydroperoxide (tBHQ)) need to undergo a further autooxidation to be able to bind to and inhibit the E3 ubiquitin ligase (i.e. KEAP1) and induce NRF2 expression. This autooxidation gives rise to reactive quinones, and importantly, is also oxygen-dependent.
Oxygen-dependent autooxidation of quinols
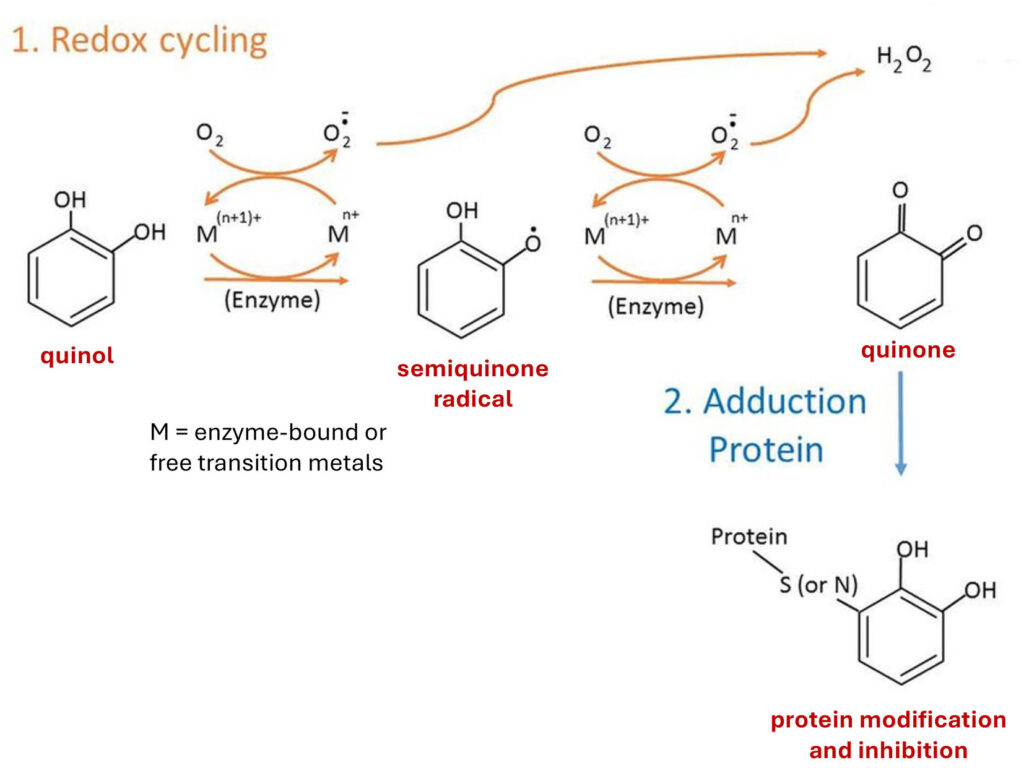
The activated quinols react with with their KEAP1 target by forming covalent bonds with cysteine residues, leading to protein inhibition.
Autooxidation of catechol quinol and covalent binding to reactive cysteines in proteins (Figure derived from Chen and Li (2019))
Given that the autooxidation of our activated quinol compounds is oxygen dependent, we compared the ability of the compounds to induce NRF2 in normoxia versus hypoxia. We noted that both 4-MC and tBHQ had a markedly reduced activity in hypoxia.
In the experiment, HEK293T cells were treated with 4-MC and tBHQ for 4 hours under normoxic (21% O2) or hypoxic (1% O2) conditions and the abundance of NRF2 was determined by Western blotting. The abundance of HIF-1α protein levels was measured as a control for the induction of hypoxia.
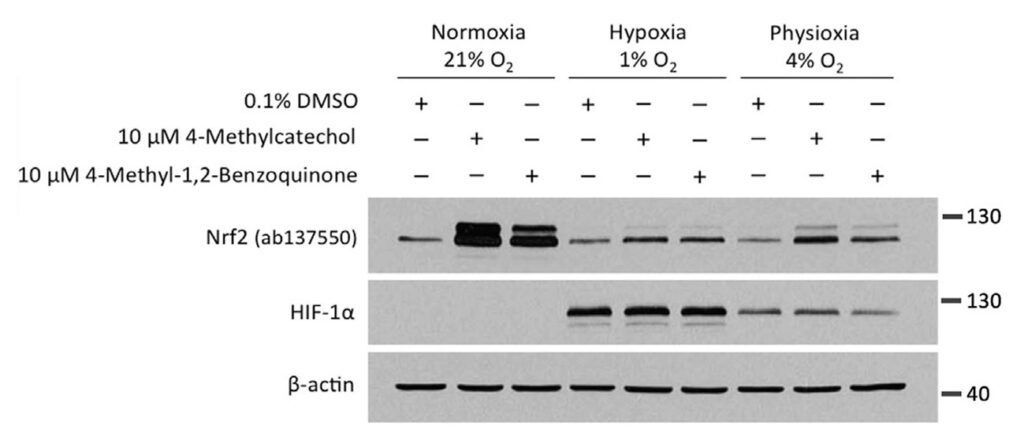
What is more, even under condition of so-called physioxia, i.e., an oxygen concentration of 4%, which is in the range of the normal oxygen concentration found in the organs in our body, both compounds had a lower activity compared to an atmospheric oxygen concentration of 21%.
We next wanted to know whether the oxygen dependence is indeed a consequence of the need of these compounds to undergo autooxidation. Towards this end, we utilised 4-methyl-1,2-benzoquinone, the oxidised active metabolite of 4-MC.
Given that 4-methyl-1,2-benzoquinone is already active and does not require oxygen-dependent autoactivation, we expected that the compound would show a similar activity in both normoxia and hypoxia. However, unexpectedly, the activity of 4-methyl-1,2-benzoquinone was also dramatically inhibited in hypoxia.
In the experiment, HEK293T cells were treated with 4-MC and its active metabolite, 4-methyl-1,2-benzoquinone, for 4 hours at oxygen concentrations of 21%, 4%, or 1%, followed by Western blotting for NRF2 and HIF-1α.
This result suggested that the low activity of 4-MC (and presumably of tBHQ) in hypoxia may not be due to a lack of autooxidation. In further support of this, when measuring the oxygen dependence of the quinol autooxidation reaction, we found that it still proceeds at a normal rate at an atmospheric oxygen concentration of 1% (i.e. in hypoxia).
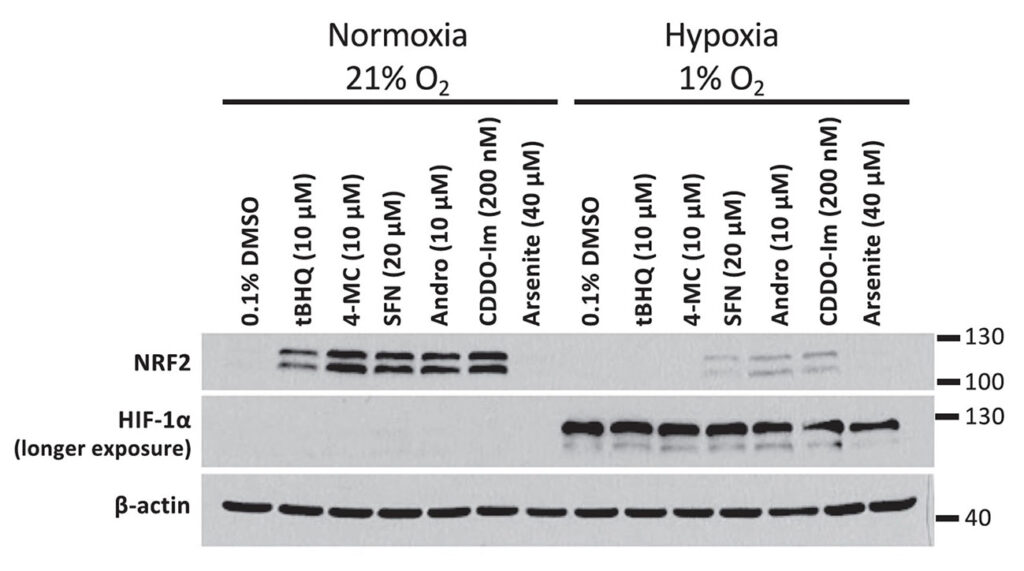
Most importantly, we found that other NRF2-inducing compounds that function via a similar mechanism as 4-MC and tBHQ, but that do not require autooxidation, also have a markedly lower activity in hypoxia.
The inhibition of NRF2 activation by hypoxia is not limited to 4-MC and tBHQ, but was also observed upon incubation of HEK293T cells with other NRF2 activators: SFN = sulforaphane, Andro = andrographolide, and CDDO-imidazole.
Taken together, we thus concluded that hypoxia must inhibit NRF2 activation by 4-MC and 4-methyl-1,2-benzoquinone as well as various other compounds via an entirely different mechanism.
Naturally, we also tried to elucidate what this mechanism is. In the course of these studies, we managed to rule out various potential mechanisms, including a change in the cellular redox state or inhibition of NRF2 translation due to lack of ATP in hypoxia or mediated by hypoxia-sensitive pathways such as mTORC1 or the unfolded protein response.
When we finally measured the rate with which the NRF2 activator compounds bind to the most reactive cysteine in KEAP1, Cys151, we found that this rate is lower under hypoxic conditions, both in cells and under in vitro conditions. The mechanistic basis for the different rates in normoxia versus hypoxia are, however, currently not clear. And so I am currently actively searching for potential mechanisms!
What are the implications of our findings? Firstly, they suggest that NRF2 activators might have low activity in tumors, which are commonly hypoxic. This is significant because NRF2 is not only beneficial and cytoprotective in normal cells, but also in cancer cells. In fact, NRF2 activation, often as a consequence of loss of function mutations in KEAP1, is a hallmark of some types of cancer, most notably lung cancer.
Therefore, our results suggest that the tumor-promoting effects of potential long-term treatment with NRF2 activators may be less of a concern when considering NRF2 activation as a treatment strategy for chronic diseases such as NASH.
Second, the results imply that it is important to take into account the effect of the oxygen concentration when evaluating any new NRF2-inducing compounds. In other words, pharmaceutical companies or labs developing new NRF2 activators would be well-advised to test their compounds at physiological oxygen concentrations present in the tissues in our body as well as at low oxygen concentrations found in tumors.
HIGHLIGHTS FOR WEEK OF 26 JANUARY – 1 FEBRUARY 2026
Goals for 2026
As discussed in last week’s post, last year I tried to focus on accomplishments and achieving ambitious goals. However, not only did I fail, but I also realised that planning to do too many things is not helpful when trying to live a happy life.
As such, my main goal this year is to live a happier and more meaningful life, by firstly committing to less. This may be difficult, given the number of things to which I have already committed. Nonetheless, I will try to maintain a focus on the things that are most meaningful for me.
Secondly, I want to become a become a more flexible person who is able to accept and embrace change, which normally goes contrary to my preference for routines and predictability.
I have always found it difficult to change my plans. Even as a child, when I had planned to play a game by myself, I found it hard to accept other kids who wanted to join in.
For all my life, I have also found it a challenge to be flexible after I start something and realise that things do not go according to my plan. For instance, during my Christmas holiday at my parents home in Germany, I encountered difficulties accessing some websites while doing my morning work routine. Instead of staying calm and waiting to re-visit the problem at a later time, I panicked and insisted on trying to solve the problem immediately, eventually switching on data roaming and hotspot Wi-Fi sharing. This led to SingTel quickly charging me over 500 dollars for the service. Thankfully, SingTel subsequently waved the charges. But the incident still caused me much self-inflicted emotional agitation.
When reading an interesting post by behavioural analyst Amy Sippl, I realised that I lack two “key characteristics of flexible thinkers”:
The ability to adapt to unexpected changes. Flexible thinkers can switch gears when plans change or routines are disrupted.
Comfort with trying new strategies or ideas. Flexible thinkers are also more willing to experiment with creative solutions and step outside their comfort zone.
Of course, it is easy to tell myself to be more flexible. However, the main difficulty is usually not figuring out what we should do, but knowing how to do it. Most people know what their weaknesses are, but they lack the knowledge of how to overcome them.
As such, I was interested in Amy Sippl’s suggestions to practice flexibility.
Firstly, there are common recommendations, such as to make back-up plans in case things do not go according to plan, and learning to not fear unfamiliar situations or new challenges by viewing mistakes as learning opportunities. However, the real problems for me typically arise when the contingency plans also fail. Moreover, the problem is not that I do not know that change is good, but that I lack the mindset to welcome and embrace change.
One way to look at uncertainty and change is that they are the normal states in the world and in our lives. There is continuous progress in the world, and this affects every part of our lives. Everything we own is constantly getting older and is getting out of fashion or demand. The same is true for our physical state and our skills and capabilities. As such, it does not make any sense to hold the expectation that what we experience today will also be true tomorrow.
What else could I actually do to become more ready to adapt to unexpected changes?
One approach is to consciously introduce change into my daily life. One example is my recent new practice to take the bus (or buses) in the morning instead of the subway. This has added uncertainty to my morning journey because unlike the train, which I can count on to arrive every 3 or so minutes, buses tend to not come at fixed times.
As an added benefit, taking the bus has made my morning rides so much more calm and enjoyable because I get to sit and watch the outside peacefully (especially because I usually go to work early when the buses are still empty!).
Amy Sippl suggests using change cards to introduce controlled disruptions. These days there are of course digital versions, such as random reminder apps that I could use as prompts to change what I am currently doing or to take a break.
After considering this approach, I eventually decided against it because I feel that it would affect my ability to enjoy my work.
What beliefs are stopping me from being able to calm down in the moment and stay with the uncertainty, or consider doing something that I had not planned?
There is my belief that without being certain, I won’t be able to think of anything else and remain productive.
There is the belief that I won’t get done what I had planned, which will spiral down to messing up all my plans.
There is also my belief that if I have planned something in one way, I have to complete it in this way.
Finally, here is the belief that by changing my plans, I will miss out on enjoyment (even though the disruption has already spoilt my ability to enjoy what I am doing.
What would it take to change these beliefs?
I could focus on thinking about how important what I had planned really is and how I can achieve the outcome in a different way.
I could focus on considering what I could potentially gain by changing my plan instead of thinking about what I might lose.
I could even try to come up with positive aspects about changing my plan.
Based on this, I came up with a Three-step plan that I will try to follow in the future:
1. How important is what I had planned?
2. If it is important, how can I achieve the goal in a different way?
3. What is a positive aspect of changing my plan?
Finally, my last goal for the coming year is to derive more joy from being, as opposed to from having and achieving stuff, and from doing good things for others, as opposed to for myself.
With regards to this last goal, I have already observed a major change as a result of having managed to resist my daily urge and habit to check out music and record websites.
This has led to two consequences. The first is the gradual disappearance of the urge to visit these websites. It reminds me of what some (although not all) Australian teenagers felt a short while after their social media ban took effect – a sense of freedom.
The second consequence is the gain of time, which has allowed me to go home on time and start the next day earlier. I hope that this trend will continue! There is some reason to believe so because positive experiences tend to reinforce newly gained habits.
HIGHLIGHTS FOR WEEK OF 19 – 25 JANUARY 2026

My 2025 goal review
I started 2025 with three main goals on my mind. My first goal was to accomplish things, in order to “have something to show for all the things that I have been learning and improving”. In fact, my original goal for the last year was to accomplish one major achievement that I could add to my CV every month.
Secondly, I wanted to increase the visibility of the things that I have been doing, specifically in the area of education. My main strategy to achieve this was supposed be to establish a LinkedIn presence.
Finally, I wanted to make progress towards my long term goals, such as coaching in athletics, health or pedagogy, and making an impact in society, for instance through outreach.
Sadly, overall I have failed in all three goals.
Although I have achieved some things (publishing a paper and finally publishing my book on education!), I got nowhere close to one achievement per month.
Publishing a research paper and especially a book is without much impact, unless I work on making them visible, which highlights why my second goal is so important. However, I ended up doing literally nothing towards this goal.
Finally, I also did not make huge progress towards my third goal, pursuing my long term goals. On closer scrutiny, I did manage to do something – I made some progress in developing and starting to implement an idea for an interesting outreach activity. Moreover, part of the reason for lack of progress in other areas were factors that were out of my control.
Nonetheless, the main reason for not achieving most of my goals last year was the lack of one important resource: Accomplishing things takes time!
Ironically, when I set my goals one year ago, I also emphasised:
“There are various ways in which I do not want to experience pursuing my goals and doing my work. I do not want to be in a constant fight with my time to get all the things done I set out for myself. I do not want to feel pressured to finish all the work and worry that I may not complete it. I do not want to feel I lost control over all the tasks that I have to do. I do not want to feel that I need to have managed to complete a long list of things in order to call the day a good day. Instead, I want to do things because I enjoy them.”
And I concluded:
“Hence, in the new year I firstly want to focus on a limited set of tasks, so that the things I do become more enjoyable. Secondly, I want to focus on enjoying the process of creating and doing things, without worrying about the achievement of perfection.”
Looking back at the past year, I felt the happiest during the semester break when I spent my days intentionally only focussing on one goal at a time. However, as the year progressed, I gradually slipped back into the habit of trying to do too many things.
The reason is likely that it is hard to resist the temptation of starting on new projects. All it takes takes is moment in which I feel excited about a new idea and start committing to it. And before I know it, I have saturated my daily capacity, leaving little or no time to regenerate and enjoy my days, and sooner or later starting to feel stressed.
As such, my biggest lesson from 2025 is how important it is to limit my daily tasks.
HIGHLIGHTS FOR WEEK OF 12 – 18 JANUARY 2026
“Going Infinite” and Feeling Depressed
I am currently reading Michael Lewis’ “Going Infinite”, which is a book the crypto company FTX and its CEO, Sam Bankman-Fried. Because I have never been interested in crypto currency, I was skeptical that reading about someone who made billions of dollars through crypto would be of great interest to me.
However, learning about Sam Bankman-Fried turned out to be fascinating. I realised (thanks to Michael Lewis’ brilliant writing) that apart from being a convicted crypto criminal, he is also a one-of-a-kind person.
Sam grew up in an academic household, with parents who identified as utilitarians, who considered that “any law should seek not to maximize some abstract notion of freedom but rather the greatest good for the greatest number”.
Through his childhood, Sam was greatly influenced by these ideas in how he viewed the world. Michael Lewis quotes Sam as saying:
“When I was about 12 years old I was first becoming politically aware and started to think through social issues. Gay marriage was a no brainer—you don’t have to be a hardcore utilitarian to see that making people’s lives miserable because they’re completely harmlessly a little bit different than you is stupid. But abortion was nagging me a bit. I was pretty conflicted for a while: having unwanted kids is bad, but so was murder.
Then Sam framed abortion as a utilitarian might. Not by dwelling on the rights of the mother or the rights of the unborn child but by evaluating the utility of either course of action.
There are lots of good reasons why murder is usually a really bad thing: you cause distress to the friends and family of the murdered, you cause society to lose a potentially valuable member in which it has already invested a lot of food and education and resources, and you take away the life of a person who had already invested a lot into it. But none of those apply to abortion. In fact, if you think about the actual consequences of an abortion, except for the distress caused to the parents (which they’re in the best position to evaluate), there are few differences from if the fetus had never been conceived in the first place. In other words, to a utilitarian abortion looks a lot like birth control. In the end murder is just a word and what’s important isn’t whether you try to apply the word to a situation but the facts of the situation that caused you to describe it as murder in the first place. And in the case of abortion few of the things that make murder so bad apply.”
And then Michael Lewis concludes: “This was how Sam figured out who he was: by thinking about things for himself …”
And this made me think of how important this skill of thinking for ourselves is if we want to improve and succeed. Sam is a genius in Math and probably other areas as well. But one does not have to be a genius to think for oneself.
It made me think of my own journey of self-improvement, that always started with thinking about things I am not happy about, trying to think of the reason(s) and trying to think of possible solutions.
But I realised that it can also be applied to external spheres of my life.
It can be applied to politics, as Sam discovered as a boy when he went to a math camp. Michael Lewis writes:
“At math camp he had conversations with other people that resembled the conversations he’d been having with himself. When other kids spoke about politics, it wasn’t to express some dumb opinion. It was to figure out the best way to model elections and predict their outcomes. When they discussed their lives and how they might live them, they actually made sense to him. Math camp kids could think their way to a belief. “And if you can’t think your way to a belief, how can you think your way to an action?” asked Sam.”
When I thought about applying utilitarian principles to my teaching, I came to an important realisation. From a utilitarian standpoint, what matters most is not trying to make everyone happy (which I tend to worry about a lot), but striving to make an impact on the class as a whole.
Sam developed his own opinions about higher education while attending MIT. University is a place with unique potential to “encourage actions that lead to the greatest good for the greatest number [of people]”. Yet, this is not what Sam found. What he discovered was that “There was very little evidence that they [the professors] were doing much of anything to change the world. Or even thinking about how to have the most impact on the world.” And so Sam became disillusioned with academia.
Although much of what I see when I look around in my surroundings fits Sam’s description, I still maintain that there are great opportunities to make an impact. It is up to each individual to recognise this possibility and take action to produce results. And this of course not only applies to academia.
Over the past weekend I experienced something I normally rarely encounter – my mood has been depressed. It was probably not a very serious case of depression, because I still managed to do what I had planned. But it was more difficult than usual, and I did not enjoy it.
And so I have been wondering where this feeling suddenly came from. There is not way to be sure, but a potential reason could be some conflicting feelings that I have been experiencing.
I have been trying to increase my visibility by starting to record some pedagogy-related videos. While I initially felt very excited about the idea of charting my teaching journey over the coming semester through videos, I soon became depressed about the quality of my videos and my perceived inability to present an engaging narrative.
There are also conflicting feelings related to the new teaching semester. On the one hand, I want to try out new things (and know that I need to in order to stay relevant). At the same time, I feel afraid that I will fail putting my new plans into practice. Indeed, there are times when I feel like someone who does not really know what he is doing.
These are typical imposter syndrome feelings, and I have read and thought about this topic a lot. I have, for instance, learned that people who have these thoughts tend to be more successful because they try harder and make more conscious efforts. However, what helps me most is trying to remember that I am not my job or my accomplishments. There is no need to prove anything. Just being, enjoying life and trying is enough.
HIGHLIGHTS FOR WEEK OF 5 – 11 JANUARY 2026
Protein design
A few weeks ago I wrote about Pinal, the generative AI based protein design tool that can predict functional proteins from language instructions.
As also discussed in the post, I was keen to try out Pinal because it seems such an amazing, almost to-good-to-be-true, tool, and also because I could potentially use it in my teaching.
Over the past month, I have managed to actually design some novel proteins using Pinal, all thanks to my amazing colleague Xavier Chee.
Xavier is a new lecturer in our department, who used his coding expertise to set up the model on my laptop and write scripts to view the output.
Seeing what difference having some basic coding skills can make prompted me to commit to learning learn some basics of Python after the next semester and try a project (such as designing a decision tree-based teaching activity with the help of ChatGPT).
Having the Pinal tool up and running, I started by trying to design proteins that bind to glutathione with high affinity, because the ability of proteins to bind to glutathione is easy to evaluate in the lab.
I then evaluated some of the predicted protein sequences in detail, using various online tools described in the original paper by Dai at al. (2024).
Firstly, I was curious on how well the 10 sequence predictions provided by Pinal align with natural glutathione binding proteins. It turned out that the first design was more than 90% identical to human glutathione S-transferase A1 (based on NCBI protein blast). However, the other nine predicted sequences exhibited very low similarities to human glutathione S-transferases or to known glutathione binding proteins from other species (see the assessment of predicted protein novelty below).
Alignment between human glutathione S-transferase A1 isoform 1 (NP_665683) and Pinal design 1 showed a more than 90% amino acid identity.
Alignment between human glutathione S-transferase A1 isoform 1 (NP_665683) and Pinal design 3 showed a low amino acid identity.
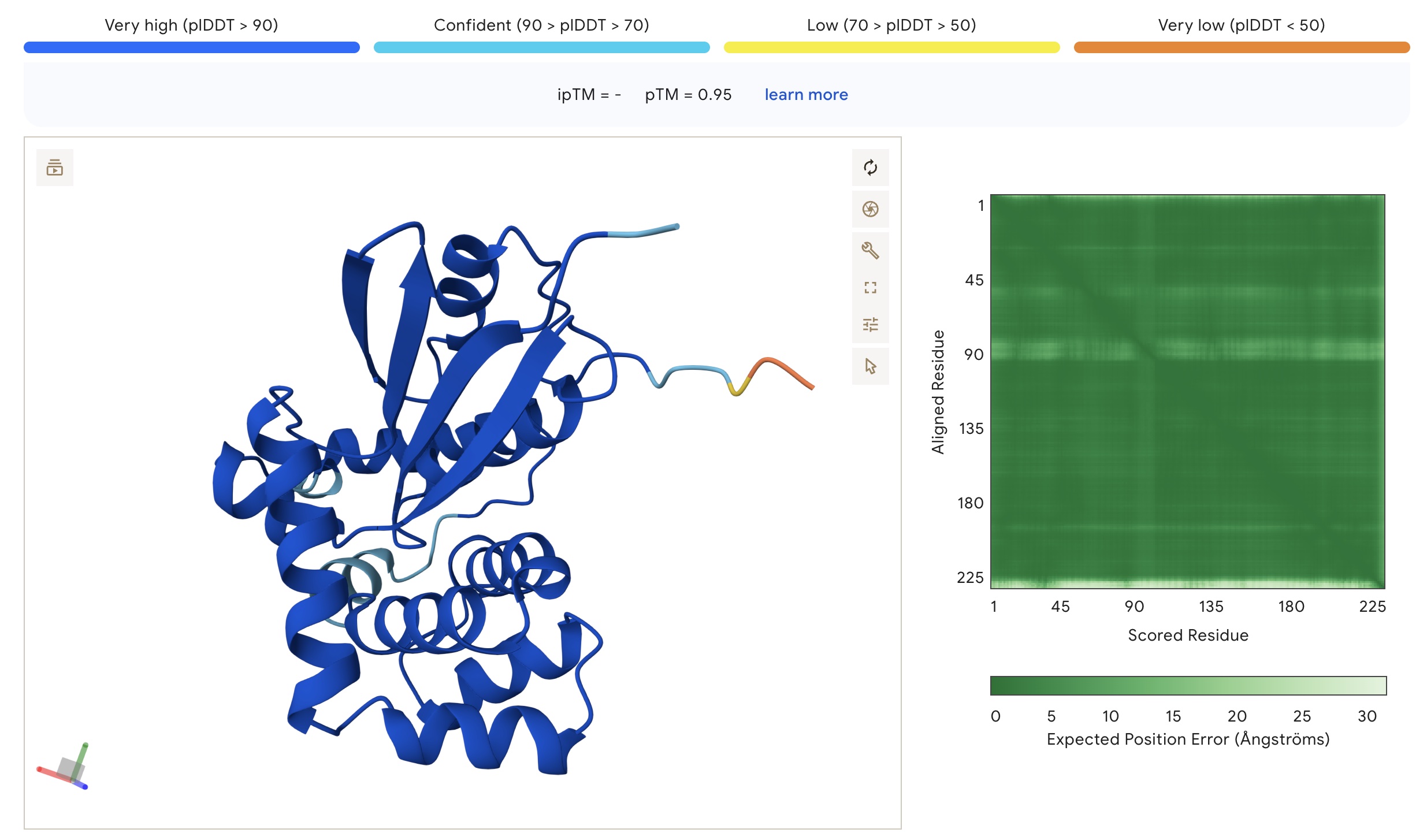
To assess protein foldability, I used AlphaFold 3, which not only predicts the protein structure based on the sequence input, but also provides measures of the likelihood with which the amino acid sequence folds into the predicted structure.
There is firstly the predicted local distance difference test (pLDDT). pLDDT measures the confidence in the local structures within the protein by assessing local distances between atoms and estimating how well they would agree with an experimental structure. As such, pLDDT provides confidence scores for individual residues and gives a value on a scale from 0 to 100 for each amino acid in a protein structure. According to Dai et al. (2024), pLDDT values above 70 are commonly considered to indicate high prediction confidence.
Secondly, there is the predicted aligned error (PAE). PAE estimates the distance error for every pair of residues. Thus, the PAE provides information about the relative accuracy between all pairs of residues. This is useful for measuring confidence in the relative positions of different protein regions. PAE is shown as a heatmap with amino acid numbers on both axes, where the colour indicates the predicted aligned error between a specific pair of amino acids. PAE values range from 0 – 35 Angstroms. According to Dai et al. (2024), PAE below 10 are commonly used thresholds indicating high prediction confidence.
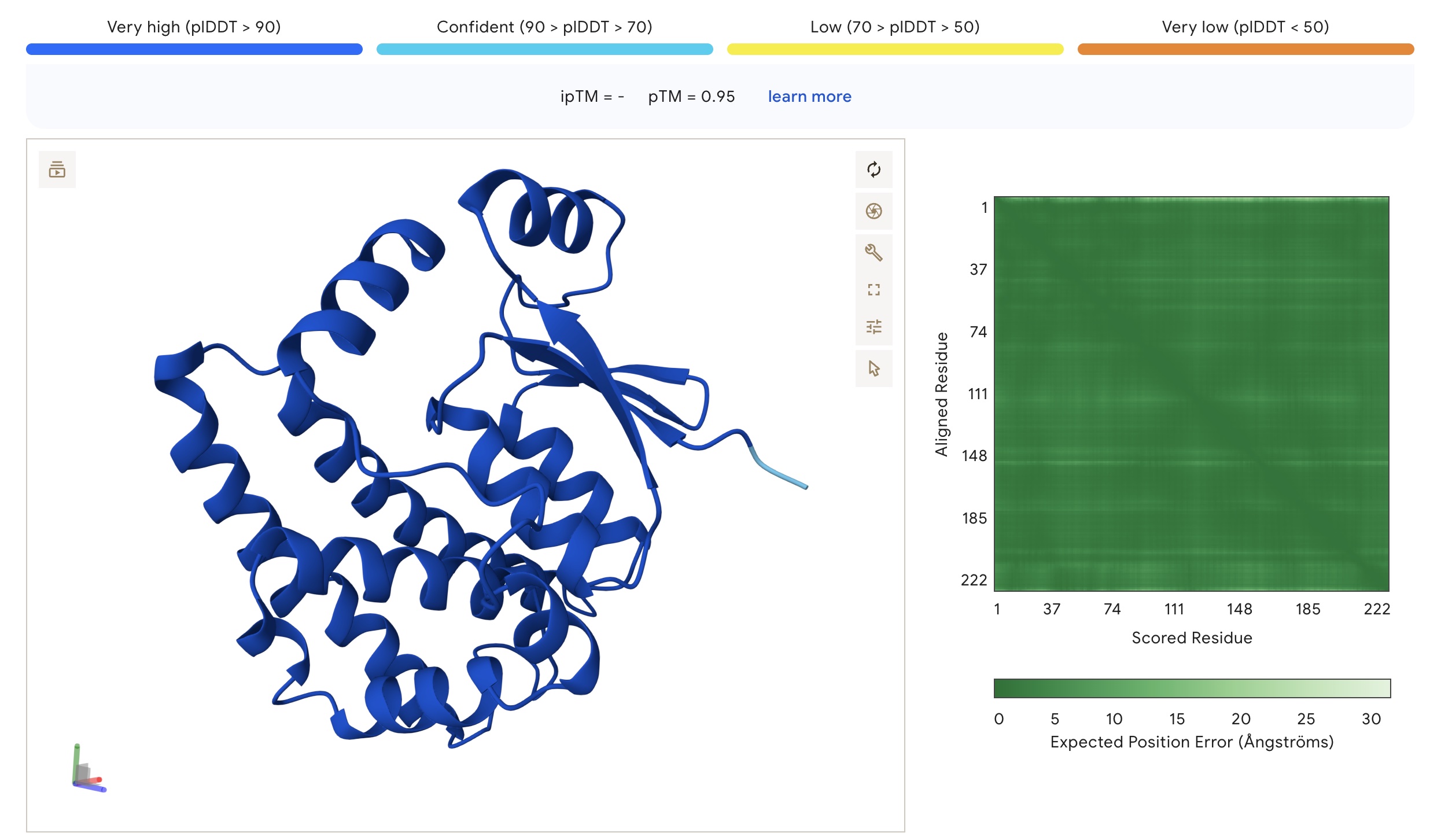
As expected, given its high homology to human glutahione S-transferase, design 1 exhibited a very high foldability, with almost the entire sequence displaying pLDDT values above 90 and PAE values below 5.
Foldability parameters of the structure of protein design 1 as predicted by AlphaFold
However, design 3 also gave pLDDT and PAE values that indicate high prediction confidence.
Foldability parameters of the structure of protein design 3 as predicted by AlphaFold
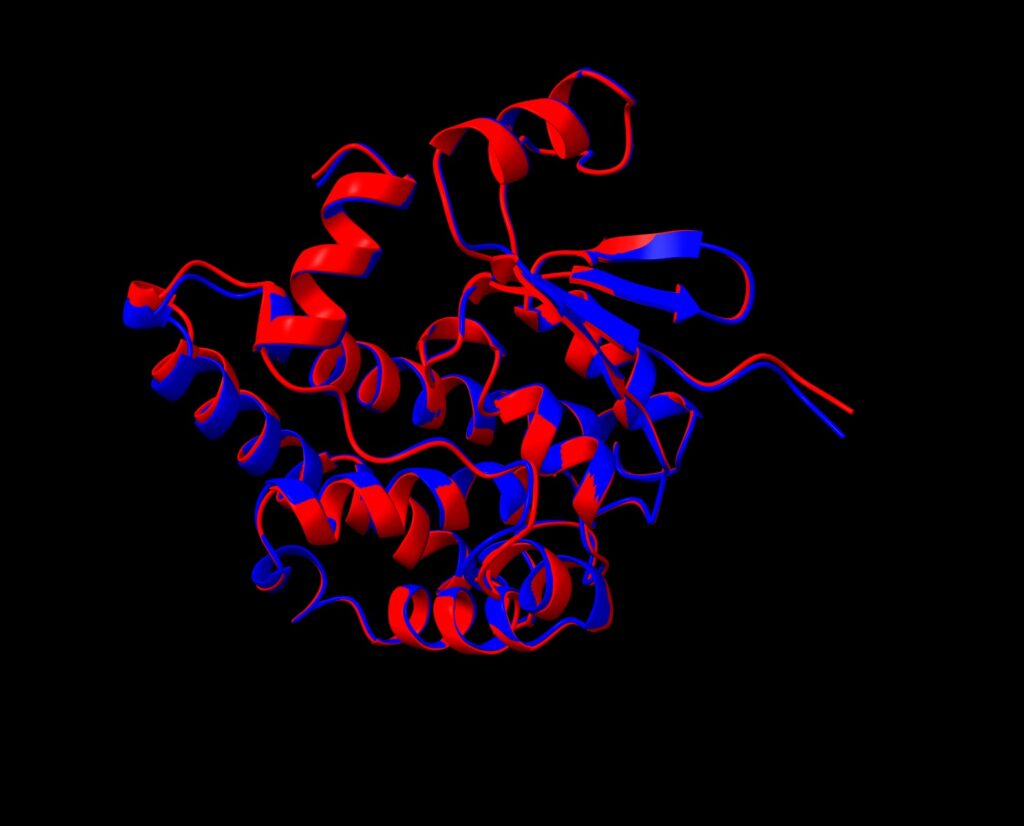
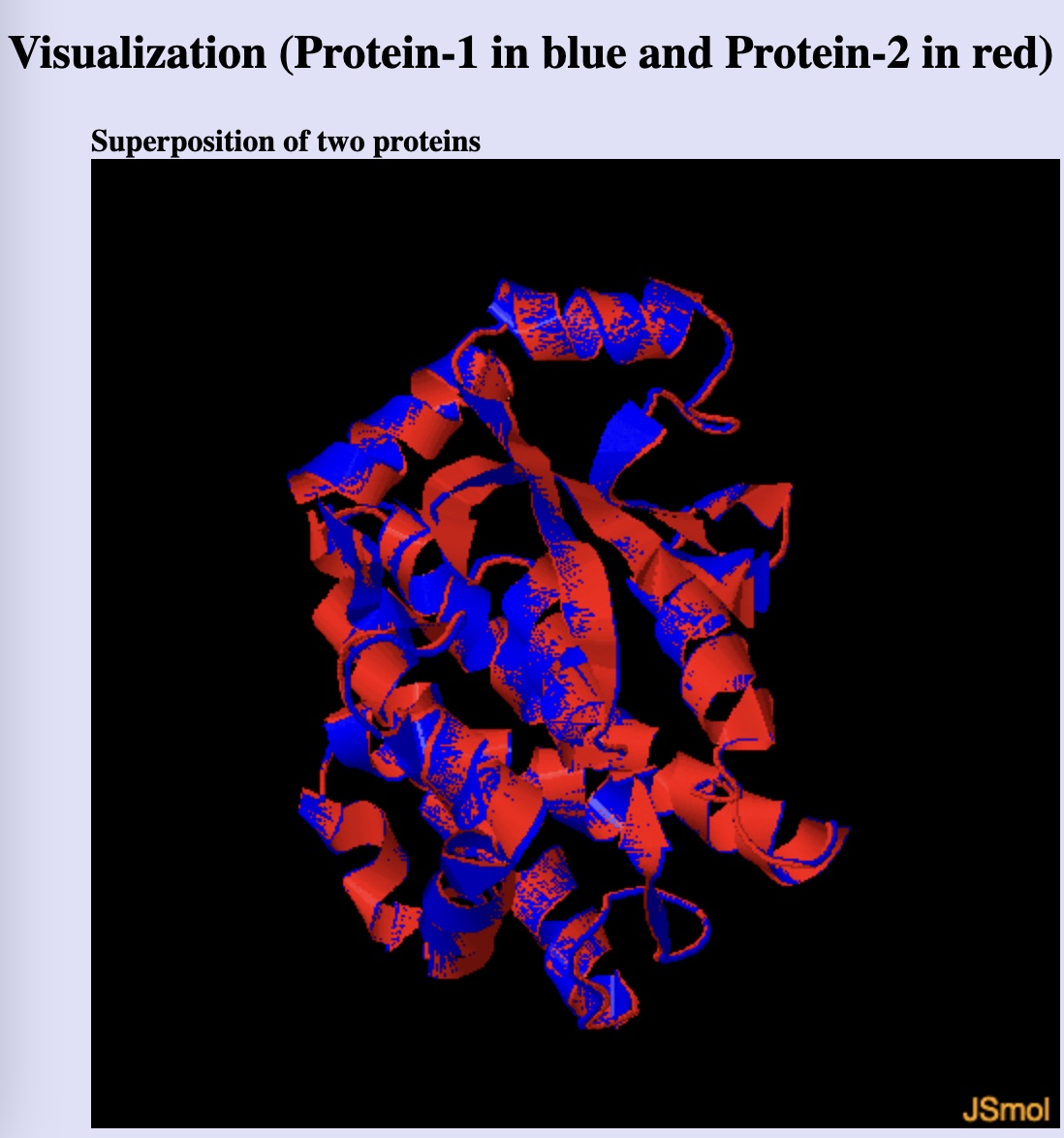
Not surprisingly, a structural alignment of human glutahione S-transferase A1 and design 1 using ChimeraX showed a nearly perfect match.
Comparison of the human glutathione S-transferase A1 predicted structure (red) and that of Pinal protein design 1 (blue) (using ChimeraX Matchmaker)
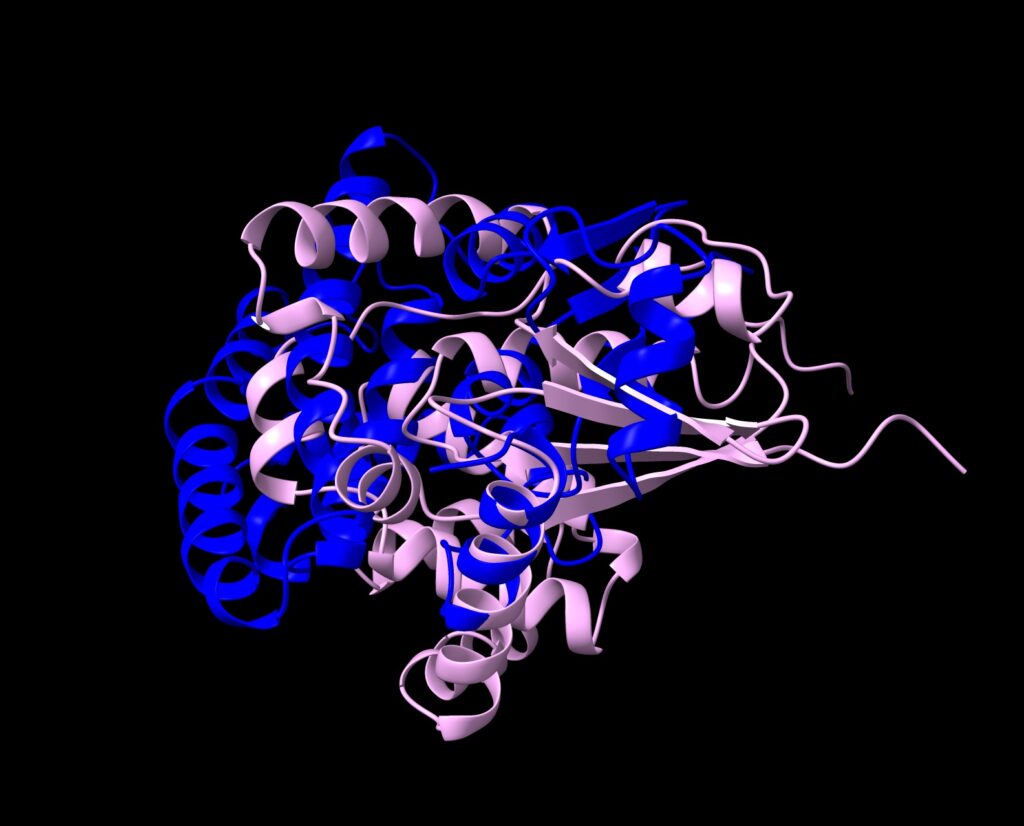
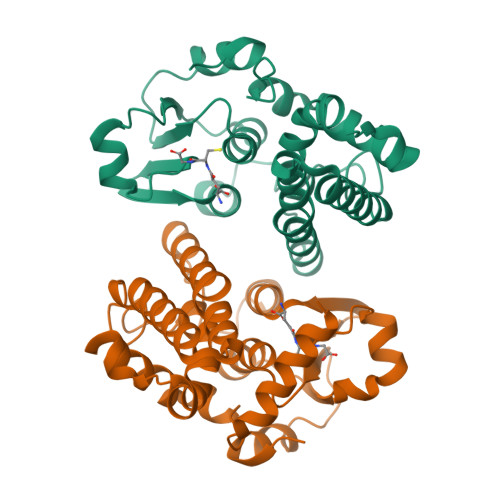
In contrast, design 3 has a very different 3D-structure from design 1, yet, exhibited good predicted foldability.
Comparison of GSH design 1 (blue) and 3 (pink)
I subsequently used designs 1 and 3 for further characterisations, first by using ProTrek.
As discussed in my previous post on Pinal, ProTrek is a tri-modal protein language model that can use either sequence, structure or text as both input and output. For instance, when using a given protein sequence as input, the tool allows one to assess the predicted function (textual output), find the closest matching structure among known proteins (structural output), and identify of the closest matching sequence of known proteins (sequence output).
When evaluating the sequence of design 1 as textual output, Protrek predicted the following function:
Design 1: Conjugation of reduced glutathione to a wide number of exogenous and endogenous hydrophobic electrophiles. Acts on 1,2-epoxy-3-(4-nitrophenoxy)propane, phenethylisothiocyanate 4-nitrobenzyl chloride and 4-nitrophenethyl bromide. Displays glutathione peroxidase activity with cumene hydroperoxide.
This result was expected, given the high homology of the design 1 sequence to human glutathione transferase.
However, design 3, despite its low sequence homology to known glutathione binding protein, gave a similar functional characterisation.
Design 3: In vitro, possesses glutathione S-transferase activity toward 1-chloro-2,4- dinitrobenzene (CDNB) and p-nitrobenzyl chloride (pNBC), and glutathione peroxidase activity toward cumene hydroperoxide and linoleic acid-13-hydroperoxide. May be involved in the conjugation of reduced glutathione to a wide number of exogenous and endogenous hydrophobic electrophiles and have a detoxification role against certain herbicides.
What is more, the matching score for design 3 was 18.89, which is even higher than that for design 1 (17.78).
Furthermore, when querying sequence to structure, highest match of design 3 was also a glutathione transferase enzyme (glutathione transferase Omega 3C from Trametes versicolor).
Finally, querying sequence to sequence confirmed that design 3 lacks close homology to known glutathione binding proteins. As the closest match to the sequence of design 3, ProTrek identified glutathione S-transferase from Arabidopsis (31.34% sequence identity).
In conclusion, based on the ProTrek characterisation, design 3 has a strikingly accurate functional annotation and is likely to function as a glutathione transferase enzyme, despite low conservation similarity in sequence and structure with to known glutathione tranferase enzymes.
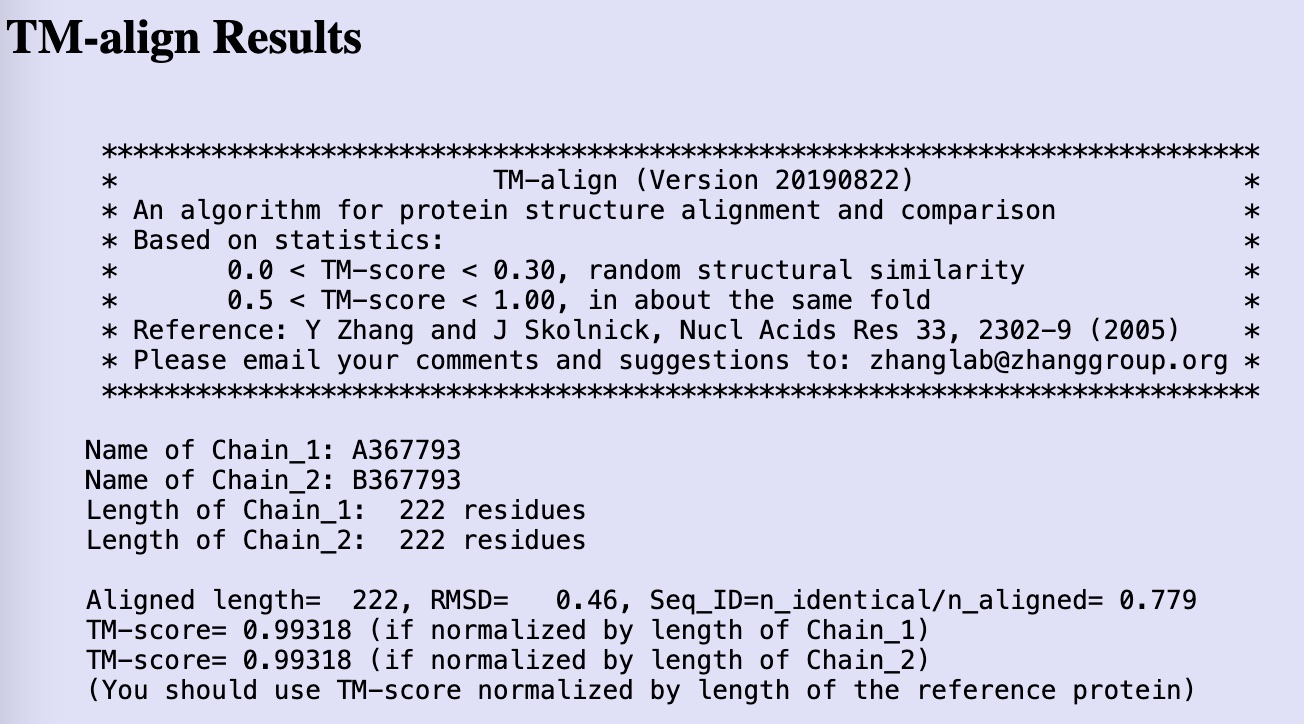
Finally, I tried to address the novelty of design using the approach described by Dai et al. 2024, in which the authors compared the structure of the designed proteins, as determined by Alphafold, to known PDB structures. To achieve this, the authors used the TM score metric.
The TM score approach was developed as early as 2004 by Zhang and Skolnick. They defined the TM score, which stands for template modeling score, as an “an algorithm for protein structure alignment and comparison”.
The website interface allows the comparison of two structures (after converting the AlphaFold cif file output into a pdb file using openbabel).
When comparing Pinal’s GSH binding protein design 1’s structure, as predicted by AlphaFold, to the structure of the sequence-wise highly similar human glutathione S-transferase A1 , I obtained, as expected, a very high TM score of above 0.99, as well as a highly homologous structure, indicative of a near perfect match.
On the other hand, comparison between GSH binding protein design 3 and its closest structural match according to ProTrek, glutathione transferase Omega 3C from Trametes versicolor, gave very low TM scores.
According to the pdb structure of Trametes versicolor glutathione transferase Omega 3C (6SRB), the protein forms a homodimer.
Pinal glutathione binding protein design 3 gave TM scores of 0.31 and 0.30 to the two chains of the homodimer, indicating a high diversion compared to its structurally most closely related protein, and thus representing a high novelty of the protein structure.
Therefore, overall, the overall bioinformatic evaluation of the design 3 sequence produced by Pinal suggests that the predicted protein is not only likely to function as a glutathione binding protein, but that it is also a highly novel protein that markedly differs from known proteins that carry out a similar function.
One caveat that I discovered, however, is that text-based instructions to restrict or define the amino acid number of the proteins to be predicted by Pinal were not successful.
What do I now do with all this?
One possibility is to use the tool in my research. However, the more exciting prospect is to utilise Pinal-based protein prediction and actual testing of predicted proteins in the lab for outreach activities. This is something that I have wanted to do for a long time.
As such, I have teamed up with the Young Educator in Science programme at NUS and their enthusiastic staff Kim Yong and Mei Ting, who are planning to develop the idea into a programme that hopefully lets MOE students experience what scientific discovery through testing new hypotheses in the lab can be like and that I can hopefully be a part of.
For older posts click HERE